Spatial Organization
Case Study 1: High-Resolution Mapping of Desmosome Organization in Human Keratinocytes
Overview
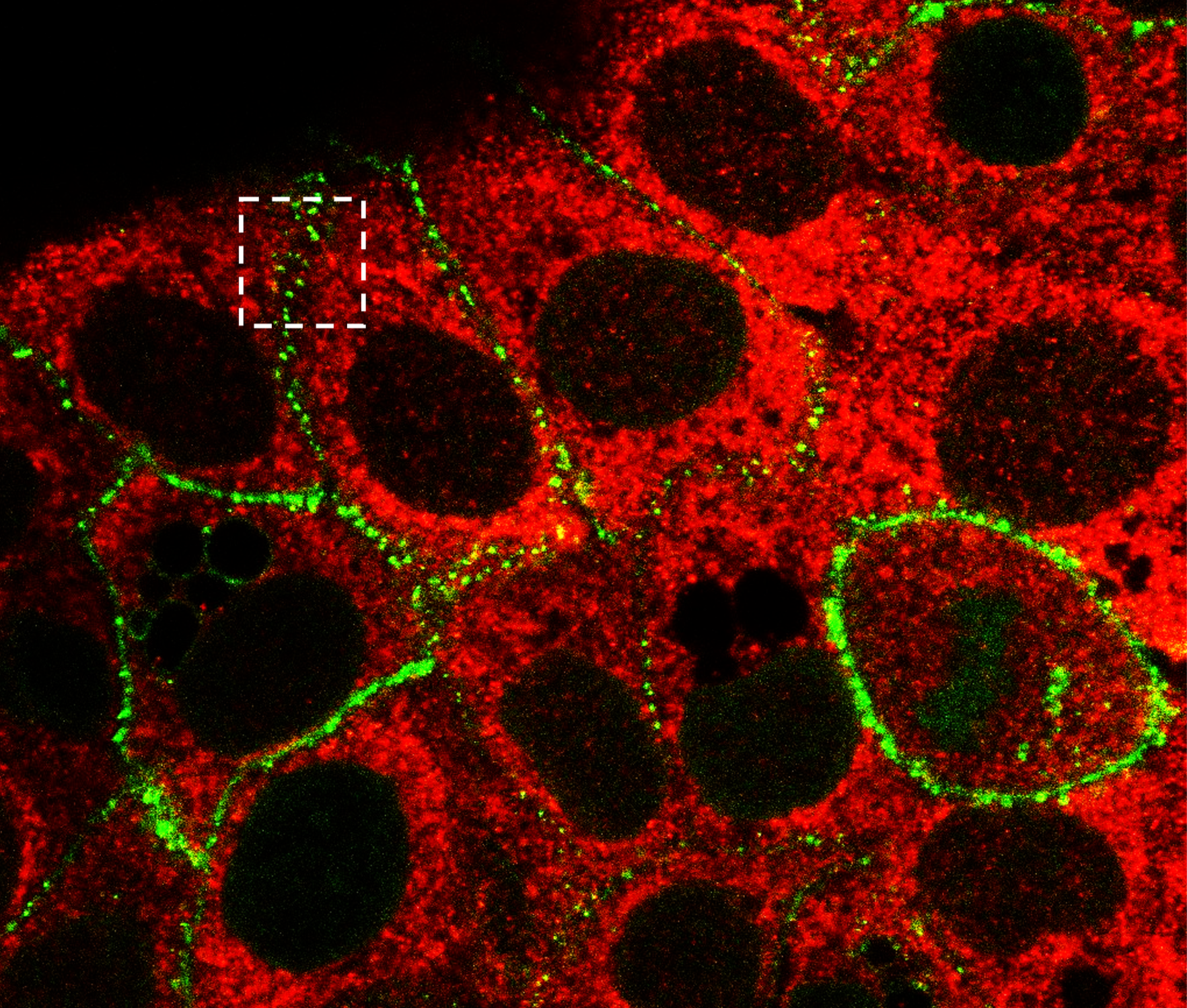
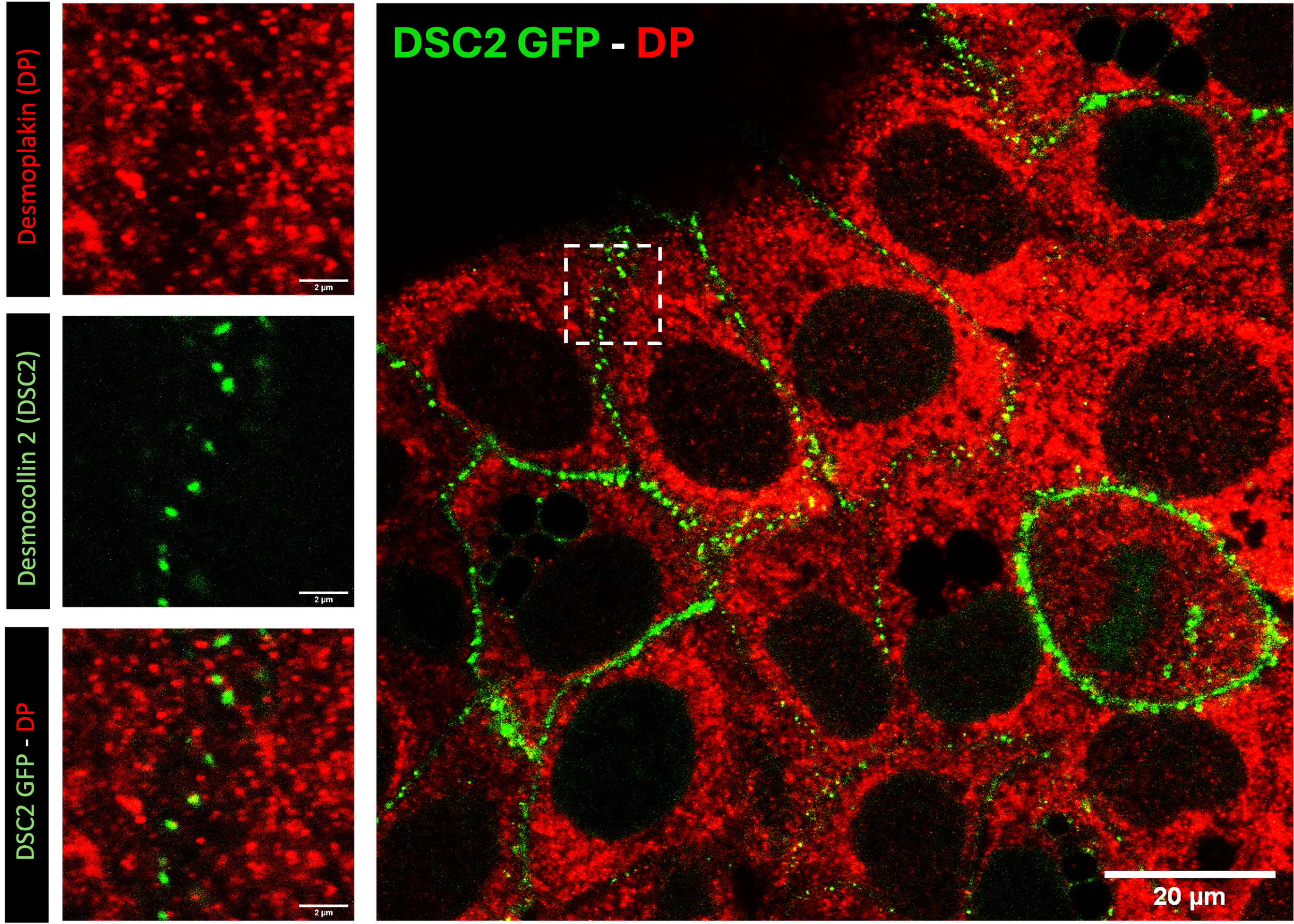
High-resolution fluorescence imaging of desmosomal structures in human keratinocytes (HaCaT cells) to investigate cell-cell adhesion and junction organization. This study demonstrates the spatial localization of desmosomal proteins and their role in maintaining epithelial integrity, with the aim of informing in vitro models that recapitulate native cell-cell interactions.
High-resolution fluorescence imaging of desmosomal structures in HaCaT keratinocytes. Desmoplakin (DP, red) and desmocollin-2 (DSC2-GFP, green) localize at cell-cell junctions, highlighting the organization of desmosomal adhesion complexes. Insets show higher magnification views of protein clustering and co-localization. This imaging approach enables detailed visualization of epithelial cell-cell interactions at subcellular resolution. Scale bars: 2 μm (insets), 20 μm (overview).
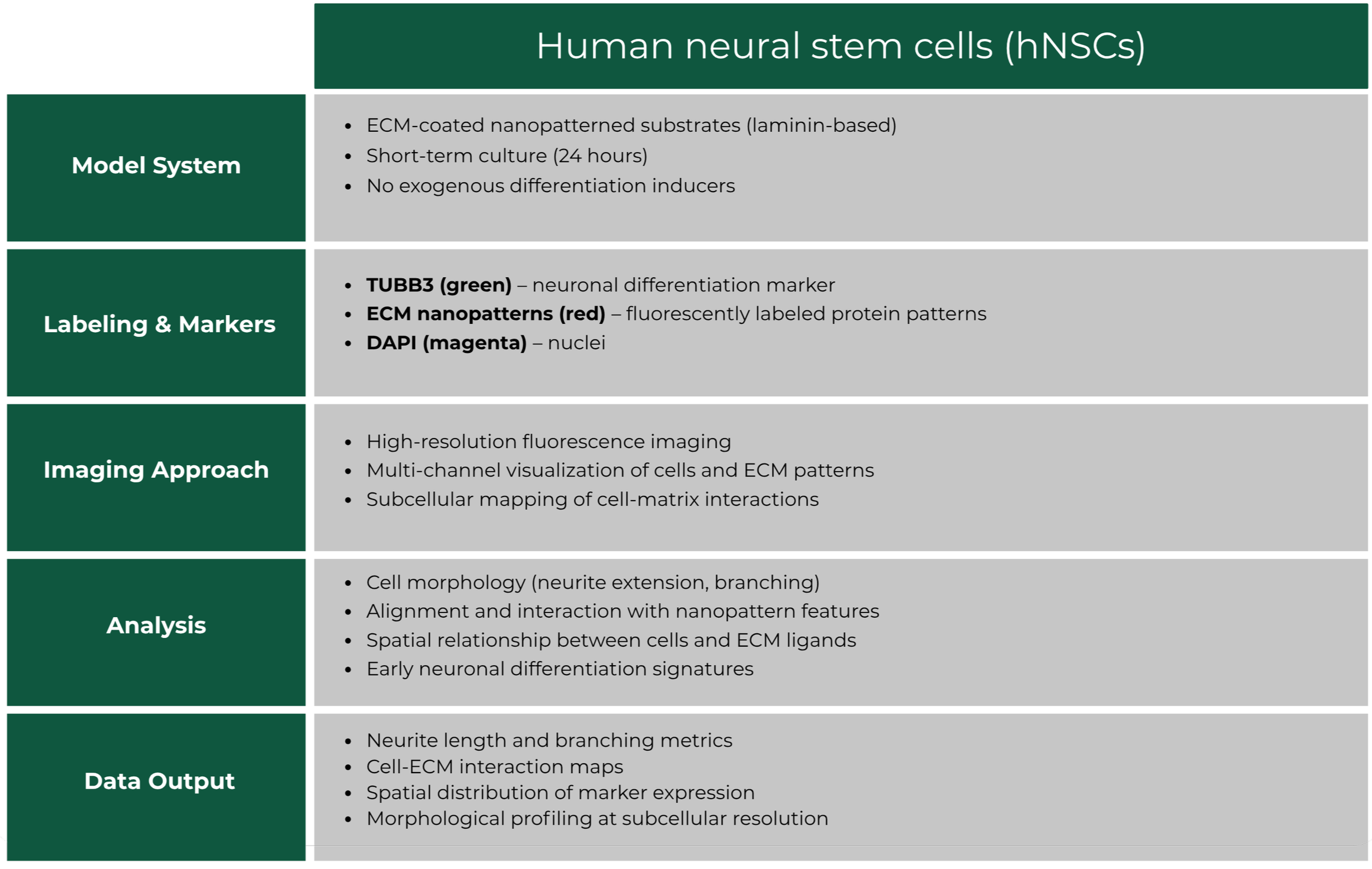
Case Study 2: Spatial Control of Neural Stem Cell Phenotype on ECM nanopatterns
Overview
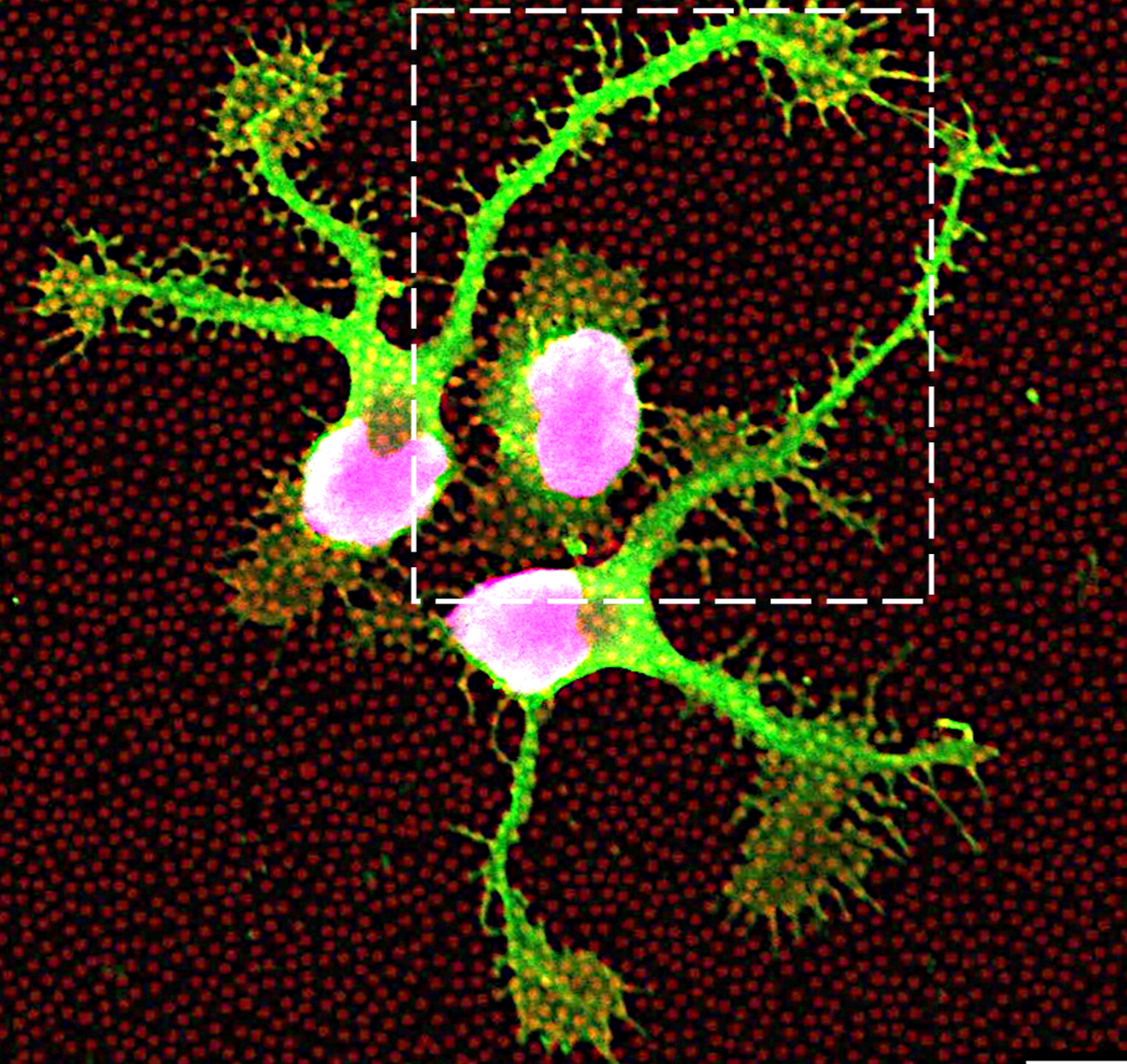
Evaluation of how nanoscale ECM patterning influences the morphology and early differentiation of human neural stem cells (hNSCs). Fluorescence imaging was used to assess neuronal marker expression and cell–matrix interactions on defined ECM nanopatterns.
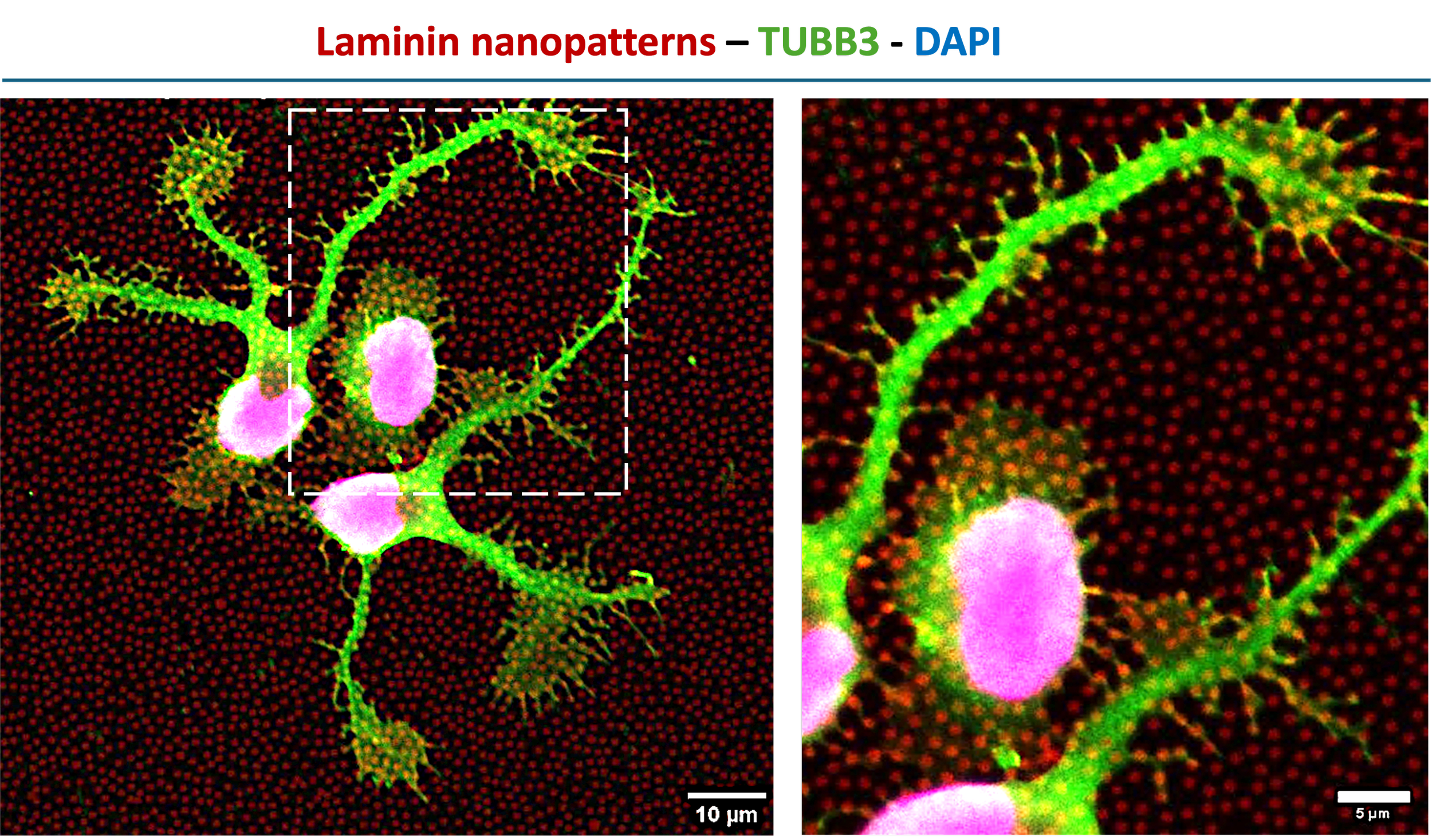
High-resolution fluorescence imaging of human neural stem cells cultured on nanopatterned ECM substrates. Laminin-based nanopatterns (fluorescently labeled, red) define the underlying microenvironment, while neuronal marker TUBB3 (green) and nuclei (DAPI) reveal cell morphology and early differentiation after 24 hours. Cells exhibit directed neurite extension and localized interactions with nanoscale ECM features, highlighting the role of matrix patterning in regulating cell behavior.
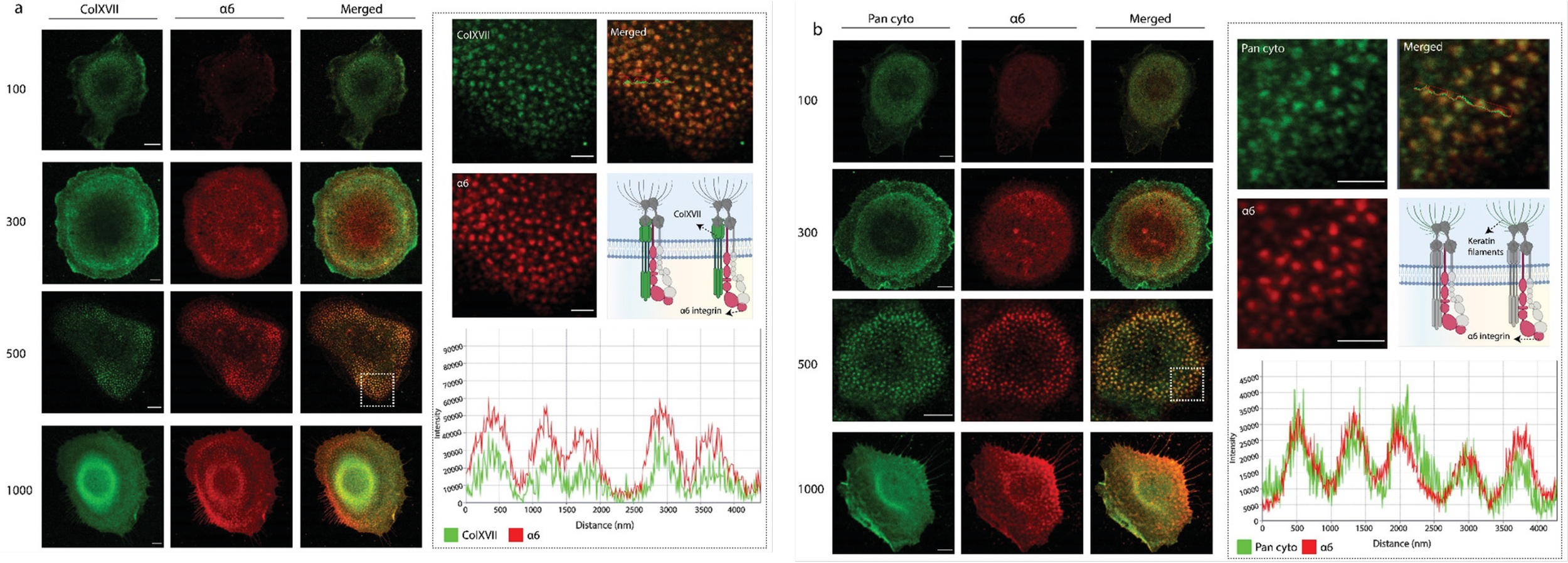
Case Study 3: Spatial Organization and Co-localization of Integrins and Cytoskeletal Markers on Nanopatterned Substrates
Overview
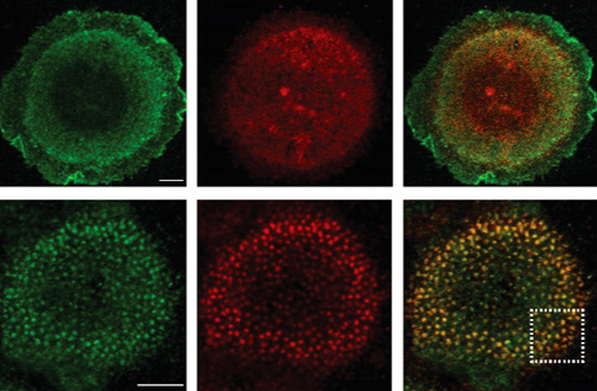
Investigation of how nanoscale surface patterning influences protein organization and cytoskeletal architecture in human keratinocytes (HaCaT cells). Multiplex fluorescence imaging was used to assess spatial co-localization between adhesion molecules and cytoskeletal components across defined nanopattern geometries.
Confocal fluorescence imaging of HaCaT cells cultured on nanopatterned substrates with varying feature sizes (100–1000 nm) after 12 hours. (a) Co-localization of Collagen XVII (green) and α6 integrin (red), highlighting organization at the cell–ECM interface. (b) Co-localization of pan-cytokeratin (green) and α6 integrin (red), illustrating cytoskeletal interactions. Merged images and intensity profiles demonstrate nanopattern-dependent spatial organization of adhesion and cytoskeletal proteins. Scale bars: 5 μm.